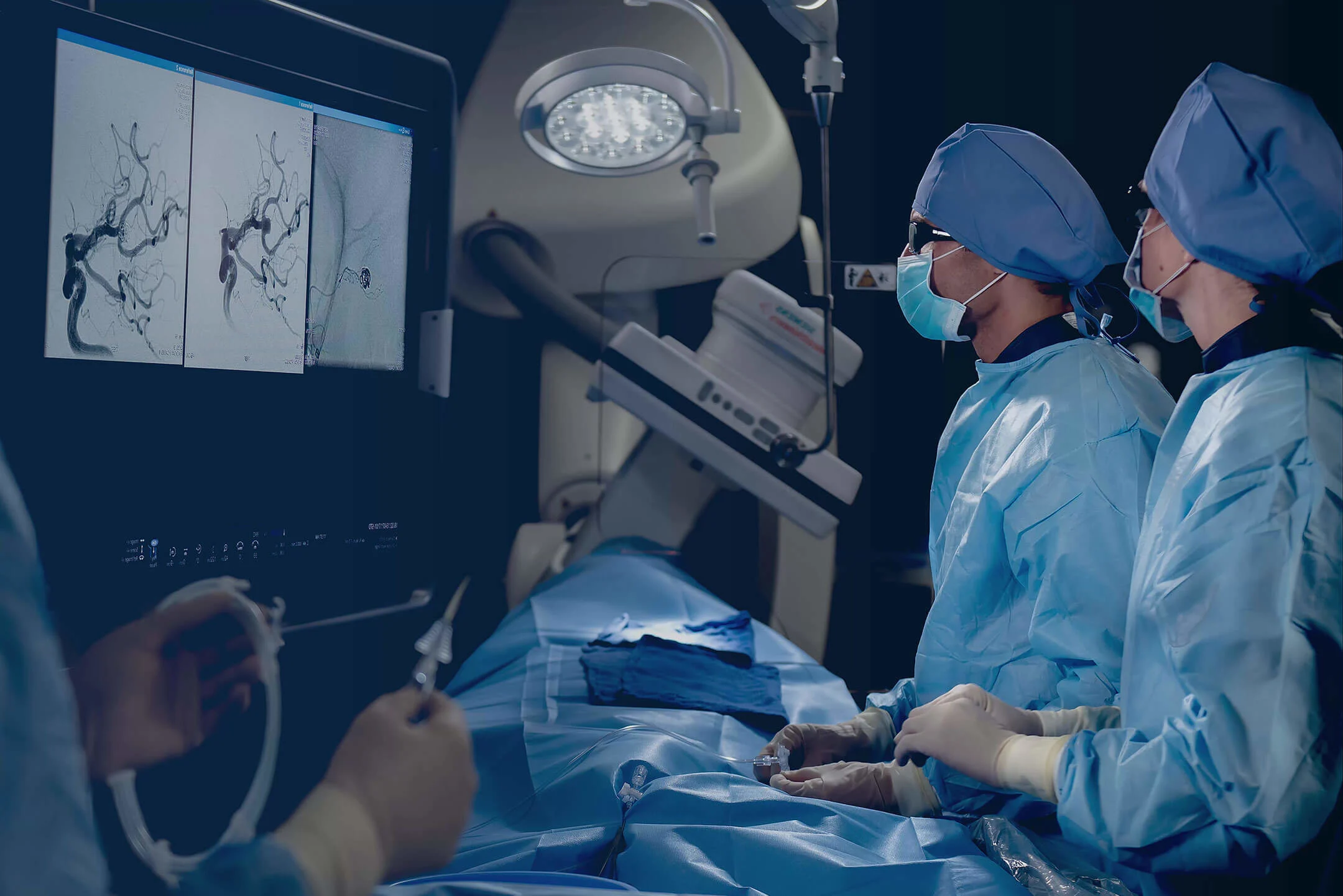
Aneurysm Therapy
A brain aneurysm is a weak, bulging spot on the wall of an artery. Our unique portfolio for ruptured and unruptured aneurysms is backed by numerous clinical studies and independent research for great performance and versatility.

Products
Our cutting-edge products lead the way in neuroendovascular therapies.
Featured Products
Aneurysm Therapy
Ischemic Stroke and Carotid Artery Disease
Neurovascular Malformations
Access Product Solutions



FDA PMA Approval
The WEB System is the first and only PMA approved device in the new category of intrasaccular flow disruptors for aneurysm embolization.
Latest Videos

Ischemic Stroke Patient Video

Tunisian Ischemic and Hemorrhagic Stroke Patient Stories

MicroVention's 25th Anniversary History of Innovations
Upcoming Events

ESMINT 2024
ESMINT (European Society of Minimally Invasive Neurological Therapy) and its Foundation (EFMINT – European Foundation of Minimally Invasive Neurological Therapy) were established in order to promote the benefits of minimally invasive neurological therapies in Europe through training and education for high-quality scientific research.

CNS 2024
Congress of Neurological Surgeons, 2024