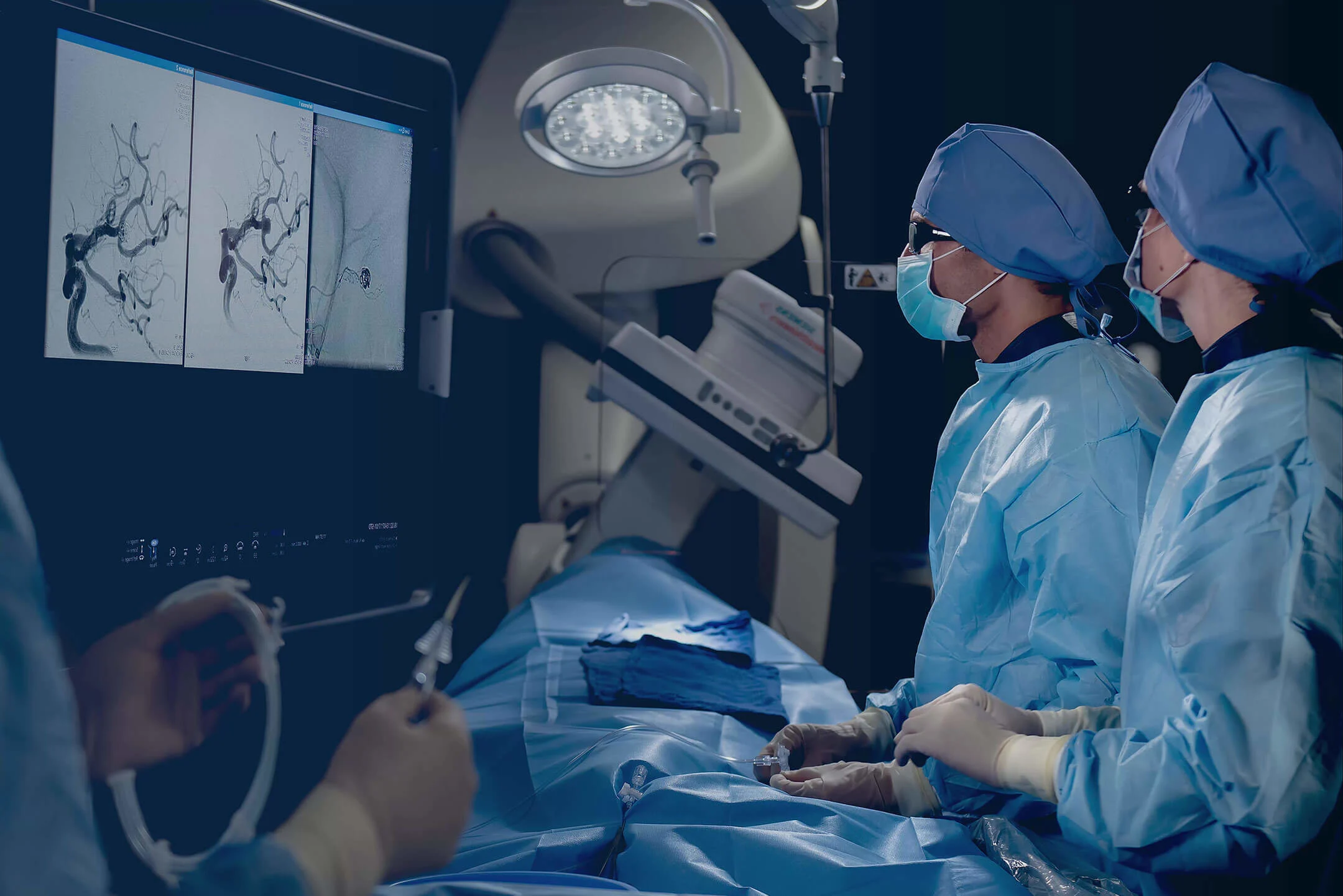
Aneurysm Therapy
A brain aneurysm is a weak, bulging spot on the wall of an artery. Our unique portfolio for ruptured and unruptured aneurysms is backed by numerous clinical studies and independent research for great performance and versatility.

Products
Our cutting-edge products lead the way in neuroendovascular therapies.
Featured Products
Aneurysm Therapy
Ischemic Stroke and Carotid Artery Disease
Neurovascular Malformations
Access Product Solutions


Latest Videos

Ischemic Stroke Patient Video

Tunisian Ischemic and Hemorrhagic Stroke Patient Stories

MicroVention's 25th Anniversary History of Innovations
Upcoming Events

May 2-4, 2024 Christchurch, New Zealand
ANZSNR 2024
The Australian and New Zealand Society of Neuroradiology is a specialty society developed for all radiologists and other clinicians with an interest in Neuroradiology, Interventional Neuroradiology or Head and Neck radiology.

May 7-10, 2024 Istanbul, Turkey
WLNC & iCure Stroke
World Live Neurovascular Conference